Comment, calculations and reasoning. First the comment.
The Question was admittedly difficult. It required knowledge of ambient pressure at
132 ft. depth, and knowing that a scuba diver inhales compressed air (21% oxygen),
not (never) 100% oxygen.
You also had to know that the value for oxygen extraction - - 25% in this case - -
only has meaning when considering oxygen content or
oxygen delivery (O2 delivery = O2 content x cardiac output).
This question was originally posted in 1999, without the answer, as a contest (the winner
received a copy of the above book). Incorrect answers received at the time included
two common mistakes:
a) mis-calculating alveolar PO2 at 132 feet depth and
b) mis-understanding that extraction is based on oxygen content, not
oxygen pressure (ie, 25% of oxygen content is extracted, not not 25% of the PaO2).
Calculations and reasoning
Step 1
132 ft of water = 4 atmospheres, plus 1 atm at the water's surface = 5atm
total. Thus, to calculate alveolar PO2, use the familiar alveolar
gas equation:
PAO2 = FIO2 (BP-47) - PCO2
PAO2 = .21 [(5x760)-47] - 1.2(40).
PAO2 = 740 mm Hg.
Step 2
The diver's alveolar PO2 is about the same as if he was inhaling
100% oxygen at sea level. It turns out that the A-a gradient goes up with increasing
FIO2; this is because nitrogen is removed from alveolar gas. However, at depth,
the alveolar nitrogen pressure is increased along with alveolar PO2, so the A-a
gradient should be normal, i.e. 5-15 mm HG. Nonetheless, we don't know what the true
A-a gradient would be, but it is reasonable to assume it should not be higher than
at sea level with 100% O2. Thus any A-a gradient could be assumed, from 10 to 100 mm Hg.
Importantly, whatever A-a gradient you assumed in this range, it would not materially
affect your answer.
Compromising, I choose 40 mm Hg A-a gradient (makes the calculations easier),
which gives arterial PO2 = 700 mm Hg.
Some answers I received placed the diver's PaO2 in the mid-600 mm Hg range,
which is perfectly acceptable.
Step 3
The next crucial step is to calculate arterial OXYGEN CONTENT, since the 25% extraction
is from oxygen content, not oxygen pressure. (This step threw most people off).
We can assume that at a PaO2 of 100 mm Hg, hemoglobin is 100% saturated.
We can also assume hemoglobin is 15 gm%, since the question stated the diver
has normal hemoglobin. Note that in this calculation the dissolved oxygen is very
important. Thus:
CaO2 = (15 x 1.34 x 1.0) + (700 x 0.003) = 22 ml O2/dl
Step 4
Because I stipulated a 25% extraction ratio, the cardiac output was not,
in fact, needed (yes, it was a distraction). Now,
25% of 22 ml O2/dl = 5.4 ml O2/dl,
so that the oxygen content in the venous blood is 16.6 ml O2/dl.
Step 5
Now you just work backwards, from the venous oxygen content to the venous PO2.
The steps are the REVERSE of what we did to calculate Oxygen content. The only difference is
that we ignore the venous PO2, since this will be low and contribute very little to the
venous oxygen content (you can consider it, and go through multiple iterations to get
the best fit, but this is not necessary).
Venous O2 content = (15 x 1.34 x Venous O2 saturation)
Venous O2 saturation = (15 x 1.34)/venous O2 content
Venous O2 saturation = (15 x 1.34)/16.6
Venous O2 saturation = 83%
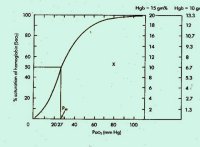
Given a venous O2 saturation of 83%, we can look up the PO2 on the standard oxygen
dissociation curve (the slight variation in pH is not important). Mixed venous PO2
(PvO2, measured in the pulmonary artery)
is therefore about 50 mm Hg. See Figure, below. (Click on figure for larger image.)
Click figure to enlarge

