Chapter 7
Acidbase balance
OUTLINE
- Hydrogen ion homeostasis
- Concept of pH
- Buffer systems
- HendersonHasselbach equation
- Does the patient have an acidbase disorder
- Calculated vs. measured HCO3-
- Acidbase nomogram
- Acidemia and alkalemia
- Acidosis and alkalosis
- Anion gap
- Primary vs. compensatory processes
- Acidbase map
- In vivo titration curve for carbon dioxide
- Base excess
- Acute vs. chronic respiratory disorders
- Acute vs. chronic metabolic disorders
- Mixed acidbase disorders
- Clinical approach to acidbase diagnosis
HYDROGEN ION HOMEOSTASIS
"Life is a struggle, not against sin, not against the
Money Power, not against malicious animal magnetism, but against
hydrogen ions."
H.L. MENCKEN
Mencken was neither a physician nor physiologist, but he knew
the importance of hydrogen ions. Enzyme systems operate at an
optimal hydrogen ion concentration ([H+]), and variation
from this optimal can markedly affect enzyme activity. For the
blood plasma, optimal [H+] is 40 nanomoles/L. As shown
in Table 71, the importance of H+ is out of proportion
to its minuscule concentration.
Strictly speaking, hydrogen ions are protons and do not exist
in the naked state in body fluids; instead they react with water
(H20) to form hydronium ions, such as H30+ and H5O2+.
For clinical purposes H+ can be used to represent these
hydrated protons. Because [H+] is so critical to enzyme
function yet the absolute concentration is small and difficult
to manipulate, the concept of pH was developed and is now universally
used to represent [H+].*
CONCEPT OF pH
The pH is the negative logarithm of the hydrogen ion concentration
([H+]):
 (Eqn 7-1)
(Eqn 7-1)
A pH of 7.4 represents a [H+] of 40 nmoles/L, or 4
x 105 moles/L (for univalent ions, mmoles/ L equal mEq/L).
By definition pH does not have units.
| Table 71. Plasma ion concentrations
|
| Ion* | nmoles/L
| mEq/L |
| H+ | 40
| 40 x 105 |
| K+ | 4,000,000
| 4 |
| Ca++ | 2,500,000
| 5 |
| Mg++ | 1,000,000
| 2 |
| Na+ | 140,000,000
| 140 |
| *K+, Potassium ion; Ca++, calcium ion; Mg++, magnesium ion; Na+, sodium ion.
|
Since pH is the negative log of [H+], the lower the
pH, the greater the [H+] and hence the greater the
acidity; the higher the pH, the lower the [H+] and
the greater the alkalinity (or the less the acidity). Use of a
logarithmic expression also means that a pH change of one whole
unit, e.g., from 7.0 to 8.0, represents a tenfold change in [H+]
.
| Table 72. pH and hydrogen ion concentration
|
| Blood pH | [H+] (nmoles/L)
|
| Acidemia |
|
| 7.00 | 100
|
| 7.10 | 80
|
| 7.30 | 50
|
| Normal |
|
| 7.4 | 40
|
| Alkalemia |
|
| 7.52 | 30
|
| 7.70 | 20
|
| 8.00 | 10
|
Table 72 shows the relationship between pH and the relative
acidity of the blood. A pH change from 7.40 to 7.30 represents
a 25% increase in blood [H+]. A similar numerical change
of conventional measurement, such as an increase in serum uric
acid from 7.3 mg% to 7.4 mg%, represents only a 1.4% increase.
The range of normal arterial pH (7.36 to 7.44) encompasses approximately
two standard deviations of the normal population; anything outside
this range is considered abnormal. Clinically, the "safe"
range for pH is approximately 7.30 to 7.52; within this range,
pH per se is not usually lifethreatening. A pH outside this
range is potentially lifethreatening because of altered
enzymatic activity and enhanced myocardial irritability, and direct
steps should be taken to return the pH to normal. Although 7.30
to 7.52 may at first seem a narrow range, it represents a [H+]
ranging from 50 to 30 nmoles/L or a change from the normal 40
nmoles/L of plus or minus 25%. A similar range for serum sodium
is 175 to 105 mEq/L!
BUFFER SYSTEMS
A buffer system counteracts the effects of adding acid or
alkali to the blood. The resulting pH change is less than if the
buffer were not present. Blood contains two basic buffer systems:
bicarbonate and nonbicarbonate. Each consists of a weak acid or
acids and their conjugate base or bases.
The bicarbonate system buffers the effects of fixed acids and
alkalies that are added to the blood; the acid component is H2CO3
and the base is HCO3-. The nonbicarbonate
system consists mainly of proteins and phosphates and serves to
buffer changes in carbon dioxide. These two systems are represented
by the equations in Fig. 71. Since the nonbicarbonate system
is a heterogeneous group of compounds, the acid component is represented
by HBuf and the base by Buf. Note that carbon dioxide is part
of an open system, since any buildup in plasma (aqueous or dissolved
CO2) can be excreted by healthy lungs.

Fig. 71. Bicarbonate and nonbicarbonate buffer
system. The two systems are in equilibrium with each other.
The bicarbonate and nonbicarbonate buffer systems are in equilibrium
with each other. Measuring the components of either system will
give the hydrogen ion concentration ([H+]) or the pH
of the blood. However, since the nonbicarbonate system is a heterogeneous
group of molecules, it is easier to measure the bicarbonate buffer
components in order to determine pH.
An extremely small quantity of H2C03 is present in the blood compared
with dissolved CO2 (approximately 1 to 400). Since
H2CO3 is in equilibrium with dissolved CO2, the latter
(measured as PaCO2) can be used as the acid component
in calculating pH. Therefore measurement of HCO3-
and PaCO2 will provide the pH.
HENDERSONHASSELBALCH EQUATION
The HendersonHasselbalch equation relates blood pH to
the components of the bicarbonate buffer system, as shown in Equation
2.
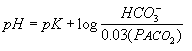 (Eqn 7-2)
(Eqn 7-2)
where pK is the negative log of the dissociation constant of carbonic
acid and has the value 6.1. The pH of the blood is equal to the
pK of the bicarbonate buffer system plus the logarithm of the
following ratio bicarbonate concentration ([HCO3-])
over 0.03 times the arterial partial pressure of carbon dioxide
(PaCO2). The constant 0.03 converts PaCO2
from mm Hg to mmoles/L. Inserting normal values gives 7.4, the
normal blood pH.
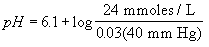 (Eqn 7-3)
(Eqn 7-3)
 (Eqn 7-4)
(Eqn 7-4)
 (Eqn 7-5)
(Eqn 7-5)
It is not necessary to memorize the full HendersonHasselbalch
equation to intelligently manage acidbase disorders. It
is important to understand that pH reflects a ratio of HCO3-
to PaCO2.
The bicarbonate buffer system is the most important of the body's
buffer systems for several reasons. This system provides the major
way to buffer the additions of fixed acid and alkali to the blood.
Since one of its components is carbon dioxide, the system is open,
i.e., the respiratory system allows for excretion of huge amounts
of carbon dioxide. Also, since carbon dioxide is readily diffusible
across all cell membranes, the results of buffering can be reflected
quickly in intracellular compartments.
Since there are three variables in the bicarbonate buffer system
(Equation 2), measurement of any two will define the third. The
body preferentially wants to maintain normal pH and does so by
altering the numerator (HCO3-) or denominator
(PaCO2) of the HendersonHasselbalch equation
as necessary.
DOES THE PATIENT HAVE AN ACIDBASE DISORDER?
It is important to recognize when a patient has an acidbase
disorder since that recognition is the first step toward diagnosis
and therapy. If any of the three variables in the HendersonHasselbalch
equation are abnormal, the answer to this question is yes. Any
acidbase derangement will be reflected in one or more components
of the bicarbonate system: pH, PaCO2, HCO3-
(see the box on p. 20 for the range of normal values).
A single abnormal component, even without knowledge of the other
two, always indicates an acidbase disorder. This is particularly
important since an abnormal HCO3- is often
found in venous blood (as part of the serum electrolytes measurement)
without a concomitant blood gas measurement. An abnormal HCO3-
value alone cannot define or diagnose an acidbase disorder
but nonetheless points to its presence. For example, an elevated
HCO3- suggests either metabolic alkalosis
or respiratory acidosis.
| Clinical problem 1 |
A 79yearold woman was hospitalized for dehydration and for cellulitis in her left leg. She received meperidine (Demerol) for pain and diazepam (Valium) for agitation. On the third hospital day she was found to be lethargic and unarousable. Review of her serum electrolytes measurements over the 3 days revealed the following information:
Day Serum HCO3-
1 35 mEq/L
2 36 mEq/L
3 36 mEq/L
No blood gas analysis was obtained until Day 3. What probably happened to this woman?
|
CALCULATED VS. MEASURED HCO3-
Incorrect therapeutic decisions can occur if blood gas
values are accepted at face value. They should always be examined
for physiologic correctness, particularly when considering acidbase
disorders, which seem prone to misdiagnosis. For example, a PaCO2
of 49 mm Hg, pH of 7.35, and HCO3- of 16
mEq/L may be interpreted as a metabolic acidosis (low pH and low
HCO3-) when in fact there is a transcription
error: the HCO3- should be 26 and cannot
possibly be 16 if the pH is 7.35 and the PaCO2 is 49
mm Hg.
Such errors can be avoided if it is remembered that HCO3-,
PaCO2, and pH must satisfy the HendersonHasselbalch
equation. If PaCO2 and pH have been measured, arterial
HCO3- can be calculated and does not have
to be measured. The HCO3- is routinely measured
as one of the serum electrolytes (on venous blood), and this measurement
can pose a problem when a comparison is made with the blood gas
HCO3-. Often, the measured venous HCO3-
does not agree with the arterial HCO3- that has been
calculated from the HendersonHasselbalch equation. When
this happens there are several possible reasons as shown in the
box below.
POSSIBLE REASONS FOR MEASURED VENOUS HCO3- NOT AGREEING WITH CALCULATED ARTERIAL HCO3-
PHYSIOLOGIC REASONS
1. The venous HCO3- measurement is actually the total CO2 content and is not identical to the plasma HCO3- calculated from the HendersonHasselbalch equation. Total CO2 content includes all the acidlabile forms of carbon dioxide, of which plasma HCO3- constitutes approximately 95%; hence the normal value for measured venous HCO3- (total CO2 content) is approximately 2 to 3 mEq/L higher than calculated arterial HCO3-
2. In critically ill or unstable patients, the pK of the bicarbonate buffer system may not be 6. 1, thus rendering calculation of HCO3- inaccurate (Hood and Campbell, 1981).
3. The venous sample may be drawn at a time different from that of the arterial sample used for blood gas analysis, and thus reflect a true change in acidbase status.
TECHNICAL REASONS
1. The blooddrawing technique may alter venous HCO3-, e.g., tourniquet placement may create a transient lactic acidosis, lowering the HCO3-.
2. The blood gases are usually measured within minutes after the arterial sample is obtained, whereas the serum electrolytes may not be measured for an hour or more after the venous sample is drawn. The venous sample's HCO3-, may change if the blood is not stored anaerobically or if its measurement is delayed .
3. If pH and PaCO2 are inaccurately measured, the calculation of HCO3- will be inaccurate as well.
4. The venous HCO3- or the arterial HCO3- may be transcribed incorrectly.
|
Note that the pK of 6.1, on which the calculated HCO3-
is based, may vary among patients. The significance of such variation
is somewhat controversial (Hood and Campbell, 1981). At most,
the variation is slight (+ 0.012 for extreme conditions) and would
not account for the wide discrepancy often found between measured
venous HCO3- and calculated arterial HCO3-.
Clinical problem 2
A 54yearoldman is hospitalized with congestive heart failure. His arterial blood pH is 7.52, PcO2 is 44 mm Hg, and HCO3- is 34 mEq/L. Measured venous HCO3- is 24 mEq/L. What is his acidbase status?
|
[Table of Contents][Previous Chapter][Chapter 7, Page 2][Chapter 7, Page 3][Chapter 7, Page 4][Next Chapter]
 (Eqn 7-1)
(Eqn 7-1)
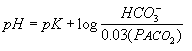 (Eqn 7-2)
(Eqn 7-2)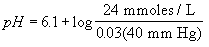 (Eqn 7-3)
(Eqn 7-3) (Eqn 7-4)
(Eqn 7-4) (Eqn 7-5)
(Eqn 7-5)