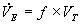 (Eqn 4-1)
(Eqn 4-1) Chapter 4
PCO2 and alveolar ventilation
OUTLINE
VENTILATION - MINUTE, ALVEOLAR, AND DEAD SPACE
In casual conversation, the word ventilation is often used by different people to mean different things. This ambiguity is often confusing for the listener, especially when terms like "hyperventilation" and "hypoventilation" are used. To prevent such confusion, it will be helpful to define certain terms at the outset.
Ventilation is a general term for the movement of air into and out of the lungs; without a preceding adjective, such as alveolar or minute, the term does not have any more specific meaning. The symbol for ventilation is V; V stands for volume and the dot for "per unit time".
Minute or total ventilation is the amount of air moved in or out of the lungs per minute. Quantitatively, the amount of air breathed in per minute (VI) is slightly greater than the amount expired per minute (VE). Clinically this difference is not important, and by convention minute ventilation is always measured on an expired sample and symbolized VE. It is useful to remember that VE is the breathing frequency (f) per minute times the tidal volume (VT, volume of tidal breath):
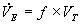 (Eqn 4-1)
(Eqn 4-1)
VE is also the sum of two other ventilations, alveolar ventilation and dead space ventilation. These and other relationships are diagrammed in Fig. 41, along with representative normal values.
Alveolar ventilation
(VA) is the volume of air breathed in per minute that (1) reaches the alveoli
and (2) takes part in gas exchange. Alveolar ventilation is often misunderstood as representing
only the volume of air that reaches the alveoli. Physiologically, VA is the volume of alveolar air/minute
that takes part in gas exchange (transfer of oxygen and carbon dioxide) with the pulmonary capillaries.
Air that reaches the alveoli, but for one reason or other does not take part in gas exchange, is not
considered part of VA (for example, air that goes to an unperfused alveolus). Such alveolar regions
lacking gas exchange constitute alveolar dead space. Clinically, the terms hyperventilation and
hypoventilation apply to alveolar ventilation only (see below).

Dead space ventilation is that part of minute ventilation that does not take part in gas exchange; it is also referred to as "wasted ventilation". Dead space ventilation (VD) includes (1) air that enters only conducting airways (referred to as anatomic dead space) and (2) air that reaches alveoli but does not exchange carbon dioxide or oxygen with the capillary blood. The combined volume of these two areas is often referred to as physiologic dead space.
Based on these definitions,
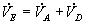 (Eqn 4-2)
(Eqn 4-2)
or
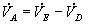
In actual practice, VE is relatively easy to measure with a spirometer
(or any device that can measure tidal volume). However, neither VA nor VD
is measured in the clinical setting; they are difficult to measure, and knowing their absolute
value is not all that helpful.
PCO2 - ITS RELATION TO ALVEOLAR VENTILATION AND CARBON DIOXIDE PRODUCTION
Although minute ventilation (VE) is easily measured, it does not provide sufficient information for assessing the adequacy of alveolar ventilation (VA), the component that affects gas exchange. The tidal volume and the respiratory rate do not give any clue as to how much air is ventilating dead space vs. alveolar space. Even if dead space ventilation (VD) and VA were measurable, the measurements would not indicate how much carbon dioxide was being produced in the body or how much VA was necessary to eliminate the carbon dioxide production.
The clinically important information can be obtained by measuring the partial pressure of carbon dioxide in the arterial blood (PaCO2); PaCO2 is the only blood gas measurement that provides information on VA. Furthermore, PaCO2 states directly, with one number, the relationship of VA to carbon dioxide production, at least at the time the sample is taken.
To understand why this is so,
consider what happens to carbon dioxide in the body (Fig. 42). Carbon dioxide is a byproduct
of food metabolism. Toxic in large amounts, carbon dioxide is literally a waste product that must be
eliminated for the body to function normally. However, carbon dioxide is also a component of the
bicarbonate buffer system, the largest of the blood buffers, and hence is an important determinant
of acidbase balance. Furthermore, PaCO2 is one determinant of arterial oxygen
pressure (PaO2) and hence plays a role in oxygenation (shown by the alveolar gas equation,
discussed in Chapter 5). Not only must carbon dioxide from metabolism be continually eliminated,
but the body must also maintain a certain level of carbon dioxide in the blood.

The total amount of carbon dioxide transported in the body is approximately 49 ml CO2/100 ml for arterial blood and 54 ml CO2/100 ml for venous blood (Fig. 42). Carbon dioxide is transported in the blood in three forms: as bicarbonate (the greatest amount), combined with hemoglobin and other proteins (carbamino compounds), dissolved. These three forms of CO2 are in equilibrium with one another, and it is the dissolved fraction in plasma that exerts the partial pressure measured as PaCO2. Normal PaCO2 ranges between 36 and 44 mm Hg; mixed venous partial pressure of carbon dioxide (PvCO2) is approximately 6 mm Hg higher.
At rest, the averagesized adult produces approximately 200 ml of carbon dioxide per minute (Fig. 42). Carbon dioxide production (VCO2) is of course a continuous process, as is oxygen consumption. VCO2 increases during exercise and declines slightly during sleep. On the average, 288 L of carbon dioxide are produced per day (1440 min x 200 ml CO2/min). That's a lot of CO2! How is this huge load eliminated while a normal PaCO2 is maintained?
Carbon dioxide elimination is accomplished by bringing fresh air into the lungs; there is no other way to excrete the body's CO2 production. (A minuscule amount of carbon dioxide, less than 1%, is excreted by the kidneys as bicarbonate.)
In the lungs, fresh air is brought in close proximity to the pulmonary capillaries, where it is separated from the mixed venous blood by a thin, gaspermeable alveolarcapillary membrane (see Chapter 1). Fresh air contains almost no carbon dioxide when it is first inhaled. In the alveoli, the partial pressure of carbon dioxide (PACO2) is practically the same as PaCO2, whatever the latter's value. Carbon dioxide is transferred across the alveolarcapillary membrane by virtue of a pressure gradient that exists between mixed venous PCO2 and alveolar PCO2, (normally 46 mm Hg and 40 mm Hg, respectively). Carbon dioxide that enters the alveolar spaces is exhaled during breathing. In a steady state, the amount of carbon dioxide produced from metabolism equals the amount eliminated by the lungs.
One of the most important physiologic relationships in all of clinical medicine can now be derived: that between PCO2 and VA. Because dead space does not take part in gas exchange, all of the expired carbon dioxide comes from the alveolar gas. Thus the amount of VCO2 excreted by the lungs equals VA times the fraction of alveolar carbon dioxide (FACO2):
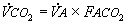 (Eqn 4-3)
(Eqn 4-3)
Since PACO2= FACO2 times total alveolar gas pressure,
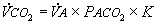 (Eqn 4-4)
(Eqn 4-4)
where K is a constant that takes into account the different units as well as the conversion of FACO2 to PACO2; 1/K has the value 0.863. Rearranging,
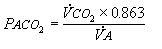 (Eqn 4-5)
(Eqn 4-5)
Equation 5 expresses the relationship between alveolar PCO2 and VA. Because carbon dioxide is a highly diffusible gas, alveolar PCO2 is assumed to be equal to arterial PCO2. Also, in the steady state, carbon dioxide excreted by the lungs equals that produced from metabolism. With these relationships in mind, it will be useful to conceptually derive Equation 5 for arterial PCO2.
Assume that carbon dioxide production is constant at 200 ml/min. Without alveolar ventilation, carbon dioxide will build up in the blood since there is no other way to eliminate it; as a result, severe acidity and death will quickly follow. The same thing will happen if there is just a small amount of VA; carbon dioxide builds up, although not quite as fast as when there is no VA.
Conversely, if VA exceeds its normal amount, excessive carbon dioxide is eliminated from the blood and PaCO2 falls. Thus as long as VCO2 is constant, PaCO2 is inversely related to the amount of VA:
 (Eqn 4-6)
(Eqn 4-6)
Now assume that the level of VA is constant at 4 L/min. If metabolism increases during exercise, for example, carbon dioxide enters the circulation at a rate greater than normal. Without a concomitant increase in VA, PaCO2 will rise. Conversely, if metabolism is slowed but VA remains unchanged, PaCO2 will fall. Thus PaCO2 is directly proportional to VCO2. Combining this observation with the one in Equation 6:
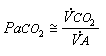 (Eqn 4-7)
(Eqn 4-7)
PaCO2 is measured in mm Hg, VCO2 in ml/min (STPD), and VA in L/min (BTPS); hence the units must be converted to mm Hg. This conversion is achieved by the constant, 0.863. Thus
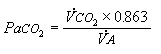 (Eqn 4-8)
(Eqn 4-8)
where
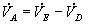 (Eqn 4-2)
(Eqn 4-2)
Of all the
relationships in this book, the one expressed by Equation 8 is among the most important.
PCO2 AND THE RESPIRATORY QUOTIENT
PCO2 is determined by the ratio of carbon dioxide production (VCO2) over alveolar ventilation (see the previous section). Carbon dioxide production is determined by several factors, including the type of foods ingested, level of exercise, body temperature, and hormonal activity (e.g., that of the thyroid hormone). Basal VCO2, approximately 200 ml/min for the average sized adult, can increase several fold during exercise (see Chapter 12).
The ratio of oxygen consumption (VO2) to VCO2 is called the respiratory quotient (RQ).
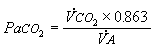 (Eqn 4-9)
(Eqn 4-9)
In a person at rest who consumes a normal diet (mixture of fats, carbohydrates, and proteins), VO2 is approximately 250 ml/min, for an RQ of approximately 0.80. When the diet consists of only carbohydrates, RQ increases to l.0 - the quantity of carbon dioxide produced equals the quantity of oxygen consumed. If only fats are eaten, the RQ is 0.71.
There is an RQ for both metabolism in the tissues and gas exchange in the lungs. In the steady state, metabolic RQ (production of carbon dioxide and consumption of oxygen by the tissues) is exactly matched by the pulmonary RQ (output of carbon dioxide and uptake of oxygen by the lungs). In fact,
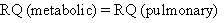 (Eqn 4-10)
(Eqn 4-10)
defines a steady state for the respiratory system. Normal ventilation is governed by the body's metabolic demands. For example, during exercise the body consumes far more oxygen and produces more carbon dioxide than at rest; as a result, minute (and alveolar) ventilation increase to meet the increase in metabolic demand.
From Equation 8 it can be seen that if VCO2 rises proportionately more than VA, PaCO2 will increase. Does this ever happen in a healthy person? For example, can one exercise to such a point that VCO2 exceeds the capacity of the respiratory system to excrete carbon dioxide? Apparently not. In fact the more a healthy person exercises, the more likely his PaCO2, will fall because of compensatory hyperventilation in response to developing metabolic acidosis (see Chapter 12).
If VCO2
increases and if VA cannot rise proportionately, PaCO2 will increase. In such
cases the cause of hypercapnia is inadequate VA for the amount of carbon dioxide presented
to the lungs for excretion. This inadequacy is discussed in the section on hypercapnia.
HYPERVENTILATION AND HYPOVENTILATION
Hyperventilation and hypoventilation are defined by PaCO2. Low PaCO2 (hypocapnia) defines a state of hyperventilation, high PaCO2 (hypercapnia), a state of hypoventilation. This terminology is often confusing to neophytes for two reasons. First, the prefixes are opposite to the level of PaCO2: hyperventilation for hypocapnia or low PaCO2, hypoventilation for hypercapnia or high PaCO2.
Second, the terms seem (at first glance) to state something about rate and depth of breathing. After all, is not someone who is breathing fast and deep "hyperventilating"? Such a person may be hyperventilating in the physiologic sense (i.e., has a low PaCO2) but then again may also be hypoventilating (has a high PaCO2). The latter could come about if most of the minute ventilation were going to dead space with very little left over for VA; this situation may arise in severe chronic obstructive pulmonary disease when there is a large amount of dead space from ventilationperfusion imbalance (see Chapter 5).
Thus hyperventilation and
hypoventilation refer to the state of VA in relation to carbon dioxide production,
a relationship that can only be known by measuring PaCO2. As used
in pulmonary medicine, the terms do not relate to rate or depth of breathing or
to any aspect of a patient's effort to breathe. Perhaps better terms are
hyperalveolarventilation and hypoalveolarventilation, but the shorter terms
seem firmly entrenched in clinical practice.
| Clinical problem 1 |
| You are called to the bedside of a patient who appears anxious and short of breath. Her respiratory rate is 24/min, and she seems to be moving a lot of air with each breath. is she hyperventilating? |
[Table of Contents]
[Previous Chapter]
[Chapter 4, Part 2]
[Chapter 4, Part 3]
[Next Chapter]