
Chapter 5
Oxygen transfer
OUTLINE
CLINICAL QUESTIONS
There are two broad clinical questions concerning the physiology of oxygenation. One question asks if oxygenation is adequate for the patient; it encompasses the patient's history and physical examination, plus measurement of his blood oxygen content, cardiac output, and oxygen delivery and Uptake. This question is discussed in Chapter 6.
The other question asks if the lungs are properly
transferring oxygen into the blood; it is answered simply by comparing
the patient's alveolararterial PO2 difference
with the expected difference.* If this difference is
elevated, the answer is no. Understanding why this is so
is the subject of this chapter.
LUNG OXYGEN TRANSFER
In Chapter 4 one gas exchange function of the respiratory system was examined: elimination of carbon dioxide. The other component of gas exchange is the delivery of oxygen from the atmosphere, through the lungs, and into the blood. Gas exchange is a passive process; there is no active transport of oxygen by the lungs.
Gas exchange is accomplished solely by diffusion
of gas from a region of higher, to one of lower, gas pressure.
Fig. 51 shows the change in oxygen pressure as oxygen moves
from the atmosphere to the arterial blood (when breathing ambient
air at sea level). Note the following (see Fig. 51):
1. The partial pressure of oxygen in dry air is the
fraction of inspired oxygen (FIO2) times the barometric
pressure; at sea level this is 0.21(760) = 160 mm Hg. With increasing
altitude, the barometric pressure falls and FIO2 remains
constant.

Fig. 51. Changes
in PO2 and PCO2 as oxygen moves from the
atmosphere to the arterial blood. All pressures are in mm Hg.
The PO2 and PCO2 are calculated using an
FIO2, of 0.21 and a barometric pressure of 760 mm Hg.
(RA, right atrium; RV, right ventricle; PA, pulmonary
arteries; PV, pulmonary veins; LA, left atrium; LV,
left ventricle.)
2. In the upper airways (nose, larynx, trachea), water vapor is added to the inspired air. Water vapor pressure is 47 mm Hg at normal body temperature; this pressure affects all dry (nonvapor) gas pressures (oxygen, nitrogen, carbon dioxide). Thus tracheal PO2 = 0.21(76047) = 150 mm Hg.
3. Inhaled carbon dioxide is negligible and can be considered zero for clinical purposes. As air travels toward the alveoli, carbon dioxide increases; PCO2 at the alveolar level = arterial PCO2 = 40 mm Hg (normal alveolar ventilation).
4. Since the lungs are an open system in continuous contact with the atmosphere, total alveolar pressure equals barometric pressure. But since inspired PCO2 is zero and alveolar PCO2 equals 40 mm Hg, the partial pressure of some other gas must fall. Water vapor does not change since it is a function of body temperature. What about nitrogen and oxygen? Nitrogen is inertit is not metabolized, and there is no net uptake or excretion. Oxygen, on the other hand, is transported into the pulmonary capillary blood at the same time carbon dioxide is entering the alveoli. The result is a fall in inspired oxygen pressure commensurate with the addition of carbon dioxide to alveolar air.
5. The fall in PO2 from the trachea to the alveoli is almost all accounted for by the alveolar PCO2 (PACO2). However, the change in PO2 does not quite equal PACO2 since more oxygen is taken up as carbon dioxide is eliminated. Under normal steady state conditions, approximately 250 ml of oxygen are added to the pulmonary circulation per minute (the VO2), while 200 ml of carbon dioxide are removed (the VCO2). The ratio of VCO2/VO2 is the respiratory quotient (R or RQ), so the normal R is approximately 0.8. Thus, as air moves from the trachea to the alveoli, PO2 will fall 1.2 mm Hg for every I mm Hg increase in PaCO2. If tracheal PO2 is 150 mm Hg and if PACO2 is 40 mm Hg, alveolar partial pressure of oxygen (PaO2) is 102 mm Hg.
6. The initial diffusing gradient for oxygen is PAO2 minus mixed venous PO2 (PVO2) = 102 - 40 = 62 mm Hg. This large gradient, plus the rapidity with which oxygen diffuses across the alveolarcapillary membrane, assures that endcapillary PO2 is almost equal to PAO2 (Fig. 52).
7. If the lungs were ideal organs, arterial PO2
would equal endcapillary PO2 and PAO2.
Normally, however, a small fraction of the cardiac output bypasses
the alveoli, either anatomically or physiologically. This shunted
blood, unoxygenated, mixes with the oxygenated blood so that arterial
oxygen pressure is always lower than endcapillary
PO2. In Fig. 5 l, PaO2 is 8 mm Hg
lower than endcapillary PO2.

Fig. 52. Alveolarcapillary
PO2 gradient. Normal transit time for a red blood cell
in the pulmonary capillary is approximately 0.75 seconds. Blood
PO2 when entering the pulmonary capillary is 40 mm
Hg. Diffusion of oxygen is rapid so that capillary PO2
reaches near equilibrium with alveolar PO2 (PAO2)
well before the end of capillary transit time. Although there
remains a very small difference between endcapillary PO2
(PCO2) and PAO2, for practical purposes,
such as when calculating shunt percentage, one can assume PCO2
= PAO2.
Based on the physiologic changes in oxygen pressure outlined above, the following observation can be made: the adequacy of overall lung oxygen transfer can be determined by comparing the oxygen pressure both in the alveolar air and in the blood entering the left atrium to normal oxygen pressure values. Since oxygen enters the blood only through the lungs, an increase in the alveolarleft atrial PO2 difference must indicate either an impairment of oxygen diffusion or an increase in pulmonary venous admixture.
It is not practical to measure oxygen pressure in
blood entering the left atrium. Instead, the difference between
alveolar (PAO2) and arterial oxygen pressure
(PaO2) must be used to assess the adequacy of oxygen
transfer. Except for the situation where some unoxygenated blood
enters the left side of the heart without passing through the
lungs, the PAO2PaO2 difference will
indicate the adequacy of pulmonary oxygen transfer. This difference
is notated P(Aa)O2. Since PaO2 is
a basic blood gas measurement and PAO2 can be calculated
from the alveolar air equation (discussed later in this chapter),
P(Aa)O2 is easily determined. If P(Aa)O2
is elevated above the normal range, the lungs are not transferring
oxygen properly.*
OXYGEN DIFFUSION AND DIFFUSING CAPACITY
Before discussing alveolar and arterial partial pressure of oxygen, it will be helpful to clarify the concept of oxygen diffusing capacity. Oxygen enters the blood by diffusion from the alveoli into the pulmonary capillaries. Diffusing capacity of the lung for oxygen (DLO2) is the quantitative amount of oxygen the lungs can transfer from the alveoli into the blood; units for DLO2 are ml 02/min/mm Hg driving pressure. If 250 ml of oxygen are transferred per minute and if the average alveolarcapillary PO2 difference is 25 mm Hg, then DLO2 is 10 ml O2/min/mm Hg.
Measurement of DLO2 is technically difficult since the gradient for diffusion changes constantly along the capillary. Using the values presented in the previous section, the initial gradient for oxygen diffusion is 62 mm Hg and the final gradient is almost zero. The decrease in diffusion gradient along the capillary is nonlinear, and the average gradient can be determined only by a complicated method employing calculus.
In practice the pulmonary diffusing capacity is measured by having the patient inhale carbon monoxide (CO) in a small enough amount that the test is harmless. The normal diffusing capacity of the lung for carbon monoxide (DLCO) is age, sex, and heightdependent and ranges from approximately 20 to 30 ml CO/min/mm Hg. The reason for using carbon monoxide is based on its properties as a gas. It is taken up so readily by hemoglobin that it is not perfusion limited; that is, no matter how fast blood flows past the lungs, carbon monoxide will combine with the available hemoglobin. On the other hand, carbon monoxide is diffusion limited so that a thickened alveolar membrane will delay its transfer into the blood.
An additional reason for using inhaled carbon monoxide is that normally very little of it is in the blood (less than 1.5% carboxyhemoglobin). Since virtually all inhaled carbon monoxide is taken up by hemoglobin, the plasma concentration and hence partial pressure of carbon monoxide remain very low (once a gas chemically combines with hemoglobin it no longer exerts a partial pressure).
For test purposes the partial pressure of plasma carbon monoxide along the entire capillary is considered zero so that CO alveolar pressure is, in effect, the pressure gradient for CO diffusion. By knowing the amount of inhaled and exhaled carbon monoxide, the mean alveolar CO pressure and the amount of carbon monoxide transferred into the blood can be calculated. Since the mean alveolar CO pressure is the gradient for diffusion, it becomes relatively easy to calculate the diffusing capacity of the lung for carbon monoxide.
There are several problems and potential pitfalls in interpretation of the DLCO test:
1. The diffusing capacity for carbon monoxide is not the same thing as DLO2, and one cannot infer any specific value for DLO2 from measurement of DLCO.
2. The diffusing capacity for carbon monoxide can be reduced by conditions other than a thickened alveolar membrane. Anemia will lower a patient's DLCO since carbon monoxide needs hemoglobin for maximal diffusion; without hemoglobin the plasma concentration would rise quickly and prevent further diffusion. Loss of alveolar membrane will also lower DLCO, and for this reason the DLCO is often reduced in emphysematous patients. Excess blood carbon monoxide, common in smokers, will also inhibit CO diffusion. Finally, ventilationperfusion imbalance can lead to a reduced DLCO.
3. A reduced DLCO does not account for clinically significant hypoxemia at rest. The diffusion reserve of the lungs is so great that DLCO (or DLO2) can be markedly reduced without lowering PaO2 very much. Diseases leading to low DLCO, such as interstitial fibrosis, cause hypoxemia by the mechanism of ventilationperfusion imbalance, not by impaired diffusion.
In practice, DLCO is helpful if it is normal, since a normal DLCO implies a normal diffusing capacity for oxygen. If the DLCO is reduced, the possible reasons mentioned previously must be considered, and one must avoid any premature conclusion about the lungs' intrinsic diffusing capacity for oxygen.
Since reduced pulmonary diffusing capacity does not
cause significant resting hypoxemia, the DLCO test is not helpful
in evaluating most abnormalities of gas exchange. There is simply
no useful correlation between DLCO and resting arterial blood
gas values. Nonetheless, the DLCO test is widely performed in
hospital pulmonary function laboratories and is considered routine
when ''complete pulmonary function tests'' are requested. Properly
performed and interpreted, the DLCO test can provide useful information.
The test is useful when treating patients with interstitial lung
disease, either as a sign of disease progression or as a sign
of response to treatment. Also, DLCO correlates with oxygen desaturation
during exercise, an application discussed in Chapter 12.
ALVEOLAR OXYGEN PRESSURE
The partial pressure of oxygen in alveolar gas (PAO2) represents an average of all the individual alveolar oxygen pressures throughout the lungs. It depends on the fraction of inspired oxygen (FIO2), barometric pressure (PB), respiratory quotient (R), and alveolar carbon dioxide pressure (PACO2). By calculating PAO2, one can obtain a good idea of what the resulting arterial PO2 should be if the lungs were functioning normally. If the difference between calculated PAO2 and measured PaO2 is increased, the lungs are not transferring oxygen properly.
Alveolar oxygen pressure is calculated using the alveolar air equation:
 (Eqn 5-1)
(Eqn 5-1)
where PIO2 is the FIO2 times the barometric pressure after water vapor pressure (47 mm Hg) has been subtracted; PACO, is alveolar partial pressure of carbon dioxide (assumed equal to Paco,); and R is the lung respiratory quotient (normally 0.8). Breathing ambient air (FIO2 = 0.21), Equation I can be abbreviated to
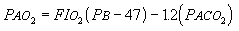 (Eqn 5-2)
(Eqn 5-2)
Physicians make several assumptions in the clinical application of Equation I (see box below). For this reason, plus the fact that there are no precise normal values for P(Aa)O2 when FIO2 is above 0.21, Equation 2 is sufficient for clinical purposes (Martin, 1986).
The assumptions listed in the box, which are routinely
made in clinical practice, render the PAO2 calculation
no more than a useful approximation; reporting PAO2
as more than a simple integer is false precision.*
| ASSUMPTIONS IN CLINICAL APPLICATION OF ALVEOLAR AIR EQUATION |
| FIO2 is accurately known. In fact, this is not always the case since face masks and other methods of oxygen delivery do not always deliver a precise FIO2.
Barometric pressure is precisely known. More often barometric pressure is approximated and is not measured. Water vapor pressure is 47 mm Hg. This value is temperature dependent and is not usually adjusted for febrile patients. PaCO2 equals PACO2. This may not always be so, especially in states of severe ventilationperfusion imbalance. R value is 0.8. Respiratory quotient may vary widely from 0.8, e.g., in patients who are hyperventilating or who have high carbohydrate diets. |
ARTERIAL OXYGEN PRESSURE
Arterial PO2 can be looked at both for what it effects or causes, and what it reflects or results from. It effects the percent saturation of hemoglobin with oxygen and thus is a major determinant of oxygen content (Chapter 6). But to look at PaO2 only in this way is to miss other important information.
Arterial oxygen pressure is a result of, and therefore
reflects, alveolar oxygen pressure (PAO2) and
the alveolar airpulmonary capillary interface. This interface
is determined by the ratios of ventilation to perfusion in the
millions of alveolarcapillary units. Oxygen enters the blood only
by passive diffusion across a pressure gradient; hence, if the
distribution of alveolar air and capillary perfusion is unchanged,
then as PAO2 increases, so will PaO2; conversely,
as PAO2 decreases, so will PaO2. PaO2
alone is insufficient to indicate the adequacy of oxygen transfer.
Without reference to PAO2 it is not known if PaO2
reflects an abnormal ratio of alveolar ventilation to capillary
perfusion or simply a reduction in PAO,.
[Table of Contents][Previous Chapter][Chapter 5, Page 2][Chapter 5, Page 3][Chapter 5, Page 4][Next Chapter]